Monkeypox: Ultimate FAQ — symptoms, last U.S. outbreak, treatment
New Jerseyans have begun hearing a good deal about monkeypox — seemingly overnight.
A day after the first U.S. case was confirmed in Boston, New York City health officials announced they are testing to confirm whether a patient there also has the rare virus.
On June 20, state health officials said New Jersey had its first "probable" case of monkeypox, which the Jersey City Health Department also confirmed.
The patient had been isolating at home in North Jersey since a PCR test result was returned on June 18, according to the Department of Health.
It is not a new virus, as the first known human case of monkeypox date back decades (1970). and many cases have been known in Africa.
In 2003, a U.S. outbreak involved people in at least six states in the first known human cases outside of Africa.
What is monkeypox?
Monkeypox belongs to the same virus family as smallpox, though it typically causes milder symptoms. Most people recover within several weeks. However, severe illness can occur in some individuals.
Here’s some of the most common questions being asked, with vetted answers from global health sources.
When was this year’s first case of monkeypox confirmed in the U.S.?
A U.S. resident tested positive for monkeypox on May 18 after returning to the U.S. from Canada.
The CDC has also been tracking multiple clusters of monkeypox reported in early- to mid-May in Canada and European countries that don’t normally report monkeypox.
Where was the 2003 outbreak in the U.S.?
In 2003, the CDC reported about 50 confirmed and probable cases of monkeypox in Illinois, Indiana, Kansas, Missouri, Ohio, and Wisconsin — traced to an African shipment of roughly 800 small animals imported from Ghana to Texas.
According to the World Health Organization, there were more than 70 human cases linked to this outbreak.
Among the mammals were six types of rodents, some of which were infected with monkeypox virus.
Some infected animals were housed at an Illinois pet store near prairie dogs, which were sold before they developed signs of the virus.
All people who became ill had direct contact with infected animals. There were no cases of human-to-human infection in the 2003 outbreak, according to the CDC.
Did anyone die in the 2003 outbreak?
No deaths were reported from monkeypox in the U.S., though at least two patients did become severely ill, one with encephalitis (inflammation of the brain).
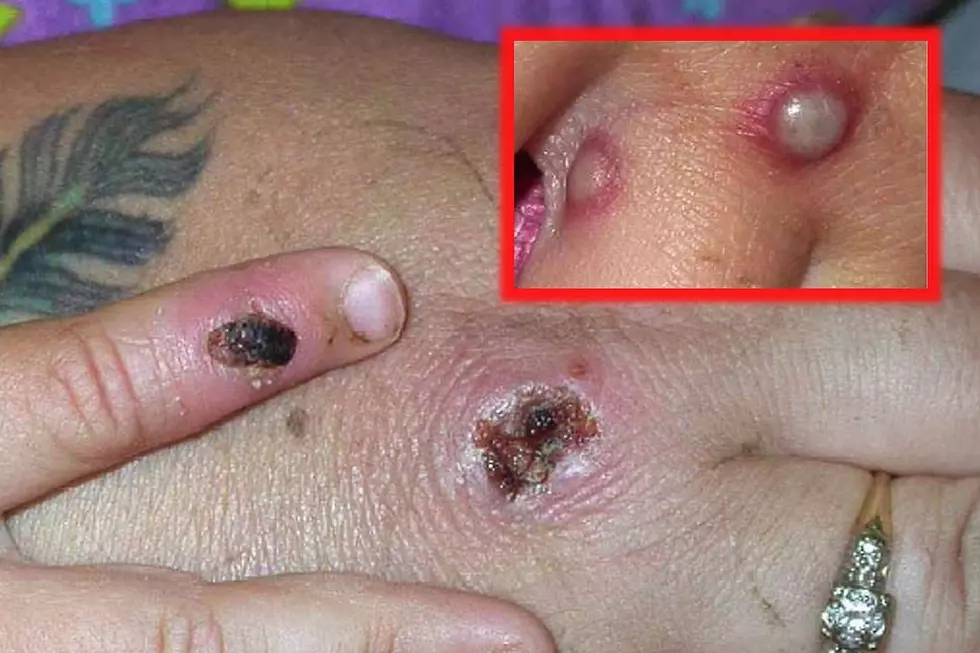
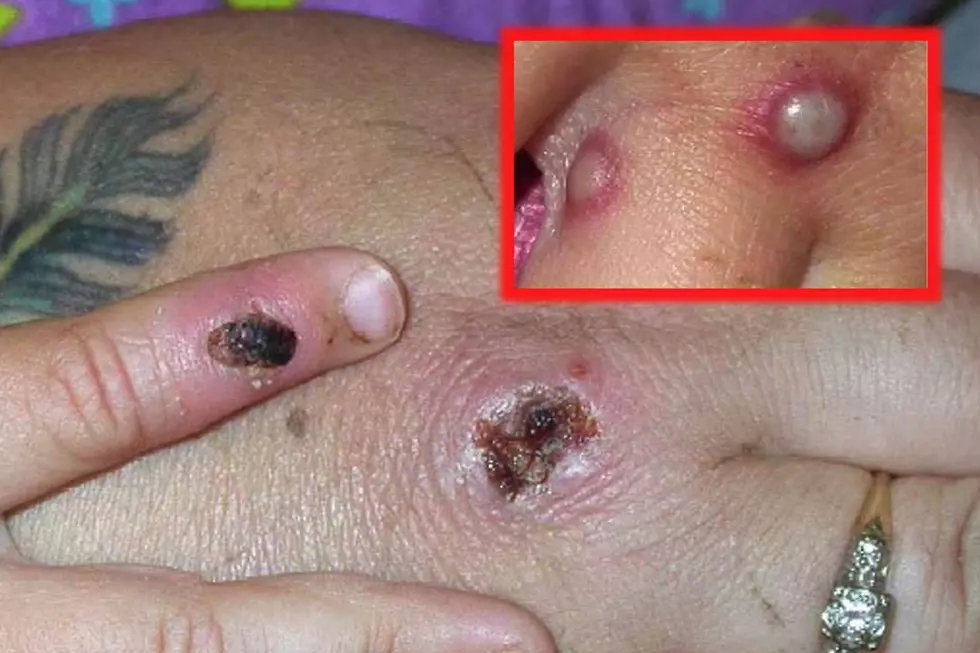
What are the symptoms of monkeypox?
Symptoms include fever, headache, backache, fatigue and chills.
Swollen lymph nodes also have been seen in a majority of patients with monkeypox.
There can also be a rash, which often begins on the face and spreads to other parts of the body.
The rash can have both raised bumps (papule) and flat, reddened areas (macule). There can also be blistering before the rash scabs over.
“An individual is contagious until all the scabs have fallen off and there is intact skin underneath,” according to health officials in the U.K.
How is monkeypox spread?
The virus enters the body through broken skin (even if not visible), the respiratory tract, or the mucous membranes (eyes, nose, or mouth).
Human-to-human transmission can happen through close contact with respiratory secretions, skin lesions of an infected person or recently contaminated objects (such as bedding).
Transmission via droplet respiratory particles usually requires prolonged face-to-face contact, according to the World Health Organization.
The incubation period for monkeypox (time from exposure to infection until the first symptoms appear) is typically 6 to 13 days — but can range between 5 to 21 days.
How often is monkeypox fatal?
Patients largely recover after symptoms that range between two to four weeks.
In recent years, the case-fatality rate of monkeypox has been 3-6%, according to WHO.
Is there a monkeypox vaccine?
As it is in the same virus family as smallpox, the existing smallpox vaccine has proven highly effective (85%) against monkeypox in the past, according to public health experts.
The smallpox vaccine was the world's first successful vaccine to be developed by Edward Jenner in 1796.
Routine vaccinations among the American public against smallpox stopped in 1972 after the disease was declared eradicated in the U.S.
The smallpox vaccine has historically involved live vaccinia virus and typically can protect you from smallpox for about three to five years. The process has continued to be different from other vaccines, as a dose is given using a "bifurcated" or two-pronged needle.
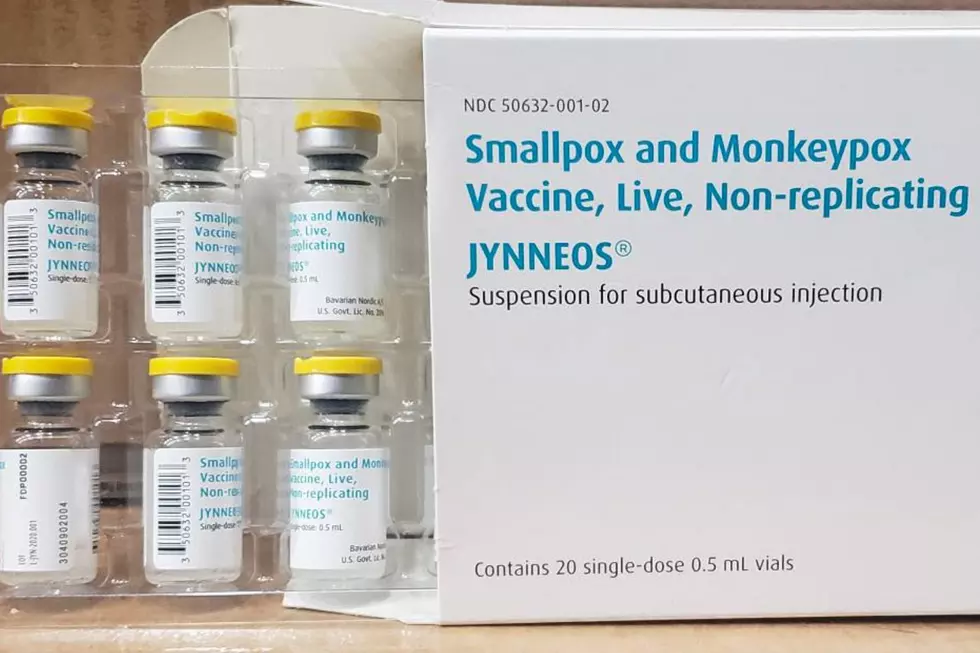
Since 2003 (the last U.S. outbreak of monkeypox), the U.S. government has been working with vaccine-maker Bavarian Nordic on boosting and improving the supply of what is known as the non-replicating smallpox vaccine.
The company's vaccine, JYNNEOS (also known as Imvamune or Imvanex), has been licensed in the U.S. to prevent monkeypox and smallpox (since 2019).
"In the event of another outbreak of monkeypox in the U.S., CDC will establish guidelines explaining who should be vaccinated, according to the CDC website.
On Wednesday, Bavarian Nordic announced it was moving forward with plans to supply a freeze-dried version of the vaccine (which has a longer shelf-life), allowing for the first U.S. doses to be manufactured in 2023 and 2024.
A federal health official told Axios on Friday that the move forward was not related to rising monkeypox concerns.
Is there a specific treatment for monkeypox?
“Monkeypox infection is usually a self-limiting illness and most people recover within several weeks,” according to U.K. health officials, though severe illness can occur in some individuals.
Up until now in the U.S., there had been no approved treatment for monkeypox virus infection. However, smallpox vaccine, antivirals and vaccinia immune globulin (VIG) can be used to treat against an outbreak, according to the CDC.
The antiviral Tecovirimat (TPOXX) has already been approved in the U.S., Canada and Europe for treatment of smallpox. (European approval also includes treatment of monkeypox, cowpox and complications from immunization with vaccinia.)
On Thursday, it was announced that the U.S. Food and Drug Administration approved an intravenous form of TPOXX for the treatment of smallpox.
Erin Vogt is a reporter and anchor for New Jersey 101.5. You can reach her at erin.vogt@townsquaremedia.com
Click here to contact an editor about feedback or a correction for this story.
Inventions you probably didn't know are New Jersey born
Gallery Credit: Judi Franco
UP NEXT: See how much gasoline cost the year you started driving
Netflix’s Most Popular English-Language TV Shows Ever
These are the best hiking spots in New Jersey
Gallery Credit: Jordan Jansson
New Jersey's new congressional districts for the 2020s
Gallery Credit: Michael Symons
More From 94.5 PST